 This resulted in a dilemma whether cathode rays are negatively charged particles or not. It was observed by Hertz well before Thomson. 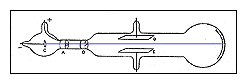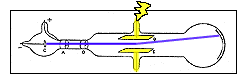
This statement became deficient when cathode rays failed to deflect in an electric field. The existence of this neutral particle, called a neutron, was confirmed in the early 1930s. The first experiment did demonstrate the behaviour of cathode rays as negatively charged particles under a magnetic field. Calculations showed that this particle should have a mass equal to that of a proton but no electrical charge. Because of this, scientists hypothesized that atoms contained still a third type of particle that accounted for these differences in mass. Thomson first measured the charge-to-mass ratio of the fundamental particle of charge in a cathode ray tube in 1897. J J Thomson giving a lecture demonstration of his m/e experiment. 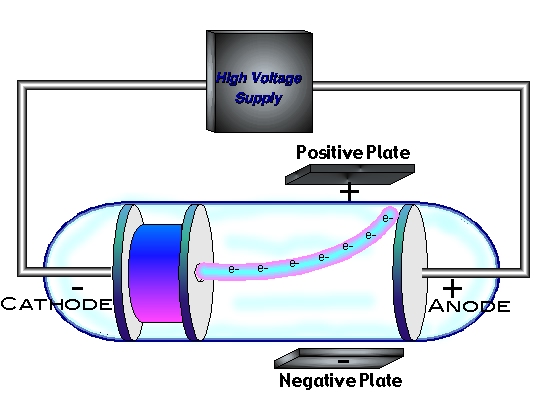
In 1896, with his student Ernest J J Thomson and the discovery of the electron Isobel Falconer St Andrews, UK Figure 1. He saw this as a way of untangling the relationship between the ether and chemical atoms. This cylinder had two slits in it, leading to electrometers, which could measure small electric charges. Upon his election Thomson began experiments on gaseous discharge. Cathode ray tube (CRT) - a vacuum-sealed tube in which electrons flow from the cathode. However, in 1910, Thomson discovered that neon consisted of atoms of two different masses. His first experiment was to build a cathode ray tube with a metal cylinder on the end. Thomson - an English physicist who used cathode rays to discover the existence of electrons in 1897. The mass of a proton was found to be only slightly less than the mass of a hydrogen atom.Īt this point, it seemed that atoms were made up of equal numbers of electrons and protons. The amount of charge on a proton and on an electron is equal but opposite, but the mass of a proton is much greater than the mass of an electron. in a vacuum with the Cathode rays in an ordinary Crookes tube was the result of. Years later, scientists determined that the rays were composed of positively charged subatomic particles called protons. The first experiment was done in order to see if the charge was separate from the cathode rays. Thomson was able to show that there rays had a positive electrical charge. When the cathode rays hit the electrometer, it measured an electric charge. 
Like the cathode rays, they are deflected by electrical and magnetic fields, but in directions opposite to the way cathode rays are deflected. These rays traveled in the opposite direction of the cathode rays. Daltonĭalton's Model of the Atom / J.J.In 1886 scientists discovered that a cathode-ray tube emitted rays not only from the cathode but also from the positively charged anode. This new model is referred to as the Plum Pudding Model. The New Model of the Atom- Thompson knew atoms were neutral, so there must be a balance of negative and positive particles. The electron is discovered, J J Thomson publishes his discovery of a subatomic particle common to all matter. Thomsons cathode ray tube experiment-an experiment in which the electron was discovered, along with its charge-to-mas. He advanced the idea that cathode rays are really streams of very small pieces of atoms. Thomson had shown that cathode rays behave as one would expect negatively charged material particles to behave. This was found to be constant regardless of the gas used in the tube and the metal of the cathode and was approximately 1000 times less than the value calculated for hydrogen ions in the electrolysis of liquids. 
J.J Thomson discovered a subatomic particle related to the cathode rays in 1897, they are known as electrons Cathode rays are negatively charged, whereas the Canal Rays are positively charged. These rays are lighter than the lightest element that is hydrogen. When investigating cathode rays using a highly evacuated discharge tube he was able to use the calculated velocity and deflection of the beam to calculate the ratio of electric charge to mass of the cathode ray. Similar to the anode rays, cathode rays also penetrate through thin metal plates. In 1897 he showed that cathode rays (radiation emitted when a voltage is applied between two metal plates inside a glass tube filled with low-pressure gas) consist of particles electronsthat conduct electricity. Amber when rubbed builds up static charge. Thomson managed to estimate its magnitude by performing experiments with charged particles in gases. Another British electrochemist John Stoney called it the "electron" from the Greek term "Elecktra" which is the Greek word for amber. When a potential difference (voltage) is applied. He called the electron, the "corpuscle" of electrical charge. Cathode ray tubes consist of an evacuated glass with two metal electrodes and rarefied gas within the glass. Thomson-Professor of Physics and Director of Cavendish Lab at Cambridge UniversityĮlectrons are distributed throughout uniformly charged positive sphere of atomic dimensions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
